Electrodeionization (EDI) Equipment
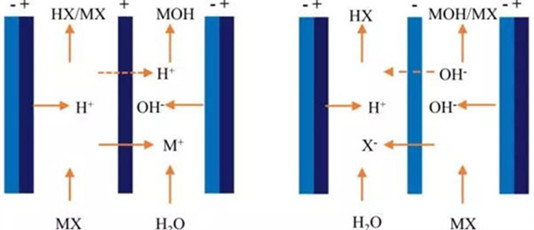
Electrodialysis (EDI) chemical water treatment equipment is a combination of an electrochemical process and a dialysis diffusion process. Driven by an external direct current electric field, it utilizes the selective permeability of ion exchange membranes (i.e., cations can pass through cation exchange membranes, and anions can pass through anion exchange membranes), causing cations and anions to move toward the cathode and anode, respectively. During the ion migration process, if the fixed charge of the membrane is opposite to the charge of the ion, the ion can pass through; if their charges are the same, the ion is repelled. This achieves the purposes of desalination, concentration, refining, or purification of solutions. Compared to reverse osmosis, another membrane separation technology introduced in recent years, electrodialysis ultrapure water equipment is cheaper but offers a higher desalination rate. Currently, the quality of domestically produced ion exchange membranes is also very stable, and operation and management are quite convenient.
Category:
Electrodeionization (EDI) Equipment
Keywords: water purification equipment, pure water equipment, design and manufacturing of purified water equipment

Consultation hotline
Contact Us
Product Description


Scope of Application:
1.Currently, EDI has a wide range of applications. It can be used for seawater desalination, seawater concentration for salt production, and refining dairy products.
2.EDI can be used for deacidification, purification, and concentration of fruit juices, as well as for producing chemical products.
3.The equipment can be used in the food, light industry, and other sectors to produce pure water, and in electronics, pharmaceuticals, and other industries for chemical water treatment.
4.EDI can be used for primary softening and desalination of boiler feed water, and for desalinating brackish water into drinking water.
5.It can be used for chemical water treatment in industries such as electronics, pharmaceuticals, chemicals, thermal power, food, beer, beverages, printing and dyeing, and coating.
6.EDI ultrapure water equipment can be used for physical and chemical processes such as material concentration, purification, and separation.
7.EDI can also be used for the treatment of wastewater and waste liquid, as well as the recovery of precious metals, such as recovering nickel from electroplating wastewater.
Inquiry Hotline:0771-3865273



|
|
|
|
|
||
 |
|
|
|
|
||
|
 |
Inquiry Hotline:0771-3865273



Basic Performance Parameters:
(1) Operating Pressure: Approximately 0.5–3.0 kg/cm²
(2) Operating Voltage and Current: 100–250V, 1–3A
(3) Power Consumption: Approximately 0.2–2.0 kWh per ton of fresh water produced
Method Characteristics::
1.Principle: In principle, an electrodialyzer is an electrolytic cell with diaphragms, capable of achieving high redox efficiency at the electrodes.
2.Co-ion Migration: The perms-electivity of ion exchange membranes is not always 100%. Therefore, a small amount of ions with the same charge (co-ions) will inevitably migrate through the membrane.
3.Concentration Diffusion: Due to the concentration difference between the concentrating and desalting chambers, a small amount of ions will always diffuse from the concentrating chamber to the desalting chamber, thereby reducing the dialysis efficiency.
4.Water Osmosis: Although the exchange membranes do not allow solvent molecules to pass through ideally, the concentration difference between the desalting and concentrating chambers can cause some solvent molecules (water) to permeate into the concentrating chamber.
5.Hydration and Electrical Double Layer: Due to ion hydration and the formation of an electrical double layer, under the influence of a DC electric field, water molecules can also migrate from the desalting chamber to the concentrating chamber.
6.Polarization Dissociation: Sometimes, under poor operating conditions, water may be forced to dissociate into hydrogen ions and hydroxide ions. These can then pass through the exchange membranes into the concentrating chamber.
7.Pressure Osmosis: Differences in fluid pressure between the concentrating and desalting chambers can force water molecules to permeate from the side with higher pressure to the side with lower pressure.Clearly, these secondary processes are unfavorable factors for electrodialysis. However, they can all be avoided or controlled by modifying the operating conditions.
Inquiry Hotline:0771-3865273

Previous page
MORE PRODUCTS
- More products -
Listen to the voice of every customer.
LISTEN TO THE VOICE OF EVERY CUSTOMER